Which of the Following Is an Oxidizing Agent
Potassium dichromate is highly electronegative in nature thus they behave as good oxidizing agents. Mn CO_5 is likely to act as an oxidizing agent.
Electrochemistry Playlist Best Chemistry Videos Class 12 Digital Kemistry Youtube Electrochemistry Redox Reactions Chemistry Lessons
Nac A Click Submit to complete this assessment.

. A F b Br c Cl d I. Start your trial now. It accepts an electron to form M n C O 5 which acquires EAN of 36 electronic configuration of Kr and is stable.
First week only 499. Save Comment David Miller. The oxidizing agents have some industrial applications like purification of water bleaching of fabrics used for the vulcanization of.
Most powerful oxidising agent to the oxidising agent is that which oxidises others and reduce themselves if they were reducing themselves it means they have high value of the reduction potential if they are having more reduction potential Hai. It is the reduced substance. Which is the stronger oxidizing agent between K2Cr207 and Cro3.
Iv D A D A. Na2SO4 sodium thiosulphate OC. 1MnCO5 2FeCO5 3Mn2CO10 4Fe2CO9 Coordination Compounds Chemistry Practice questions MCQs Past Year Questions PYQs NCERT Questions Question Bank Class 11 and Class 12 Questions NCERT Exemplar Questions and PDF Questions with answers solutions explanations NCERT reference and difficulty level.
M nCO5 e M nCO5 Less stabl e Oxidizing Agent 35 1 36. Strongest reduction agent is. A 1 and 2 only B 1 and 3 only C 2 and 3 only D All of 1 2 and 3 E Neither 1 2 or 3.
The CO ligand will intetact through its sigma and pi orbitals with all valence orbitals one s three p and five d orbitals hence the 18 electron rule. Chemistry questions and answers. The CO ligand will intetact through its sigma and pi orbitals with all valence orbitals one s three p and five d orbitals hence the 18 electron rule.
Etc are the examples of weakest oxidizing agents. KMnO 4 H 2 S HCl KCl MnCl 2 H 2 O S 8. Which of the following reagents is not typically viewed as an oxidizing agent.
MnCO_5 is likely to act as an oxidizing agent. The strongest oxidizing agent in the list is F_2 followed by H_2O_2 and so on down to the. Common examples of oxidizing agents include halogens such as chlorine and fluorine oxygen and hydrogen peroxide H 2 O 2.
F is the strongest oxidizing agent due to its highest electronegativity and stable configuration attained by it after accepting electron. Cause an increase in the oxidation state of the substance by making it lose electrons. Cl 2 has a larger positive value of E 0 than Br 2Thus Cl 2 is a stronger oxidizing agent than Br 2.
First determine the oxidation number of every atom on both sides of the arrow. I A B A B. Let 2sin2x3sinx20 and x2x2Which of the following.
Which of the following is an oxidizing agent. Heres a typical table of standard reduction potentials. Which of the following are typically TRUE of an oxidizing agent.
Bolivianouft and 13 more users found this answer helpful. Which of the following reagents is not typically viewed as an oxidizing agent. Oxygen hydrogen peroxide and halogens are the common oxidizing agents.
A less stable M n C O 5 e more stable as EAN of Mn36Kr M n CO 5. Weve got the study and writing resources you need for your assignments. SO24SO42aq Liaq Iaq MnO4-aq ClO2s close.
Carbonyl complexes are most stable when there are 18 around the central transition metal. Solution for Which of the following is the strongest oxidizing agent. On the basis of given reactions the correct order of increasing oxidising ability of A B C and D is.
Which one of the following is the best oxidizing agent A 2 H B Na C 2 O D Li E from PCB 3233 at University of Central Florida. Which of the following is the strongest oxidizing agent. Atom compound or ion which gives oxygen to another substance or removes hydrogen from a substance is called oxidizing agent.
Compound that gets reduced is generally called oxidising agent. Iii C B N o r e a c t o n. A PCC b H 2 CrO 4 c KMnO 4 d H 2 O 2 e LiAlH 4 CHEM244 Fall 2021-22 Midterm Exam 27.
Which Of The Following Is The Strongest Oxidizing Agent. You rank oxidizing agents according to their standard reduction potentials. M n C O 5 acts as an oxidizing agent.
Flourine is the most electronegative hence forming F is the most energetically. More stable as EAN of Mn 36 kr Related Questions. Determine the oxidizing and reducing agents after answering the following questions.
Here zinc is oxidized to Zinc sulfate and copper sulfate acts as an oxidizing agent. What is an Oxidizing Agent. Ii C D N o r e a c t o n.
An oxidizing agent often referred to as an oxidizer or an oxidant is a chemical species that tends to oxidize other substances ie. Fluorine is a most powerful oxidizing agent because its reduction potential tendency to get reduced is very high at 25 V. The maximum oxidation state for potassium permpermanganate is 7 thus they act as a good oxidizing agent.
0 followers 0 following Joined September 2020. Please log inor registerto add a comment. Correct choice is b CrO3.
Carbonyl complexes are most stable when there are 18 around the central transition metal. Which of the following is an oxidizing agent. HOCI ОА KMnO4 ОВ OC H202 02 OD KCI The product isolated in the Oxidation Reaction was dried using ОА Naoc OB.
Chromic anhydride CrO3 dissolved in glacial acetic acid is sometimes used as an oxidizing agent.

Rank The Following In Increasing Ability As Oxidizing Agents In 2022 Oxidation Low High Ranking

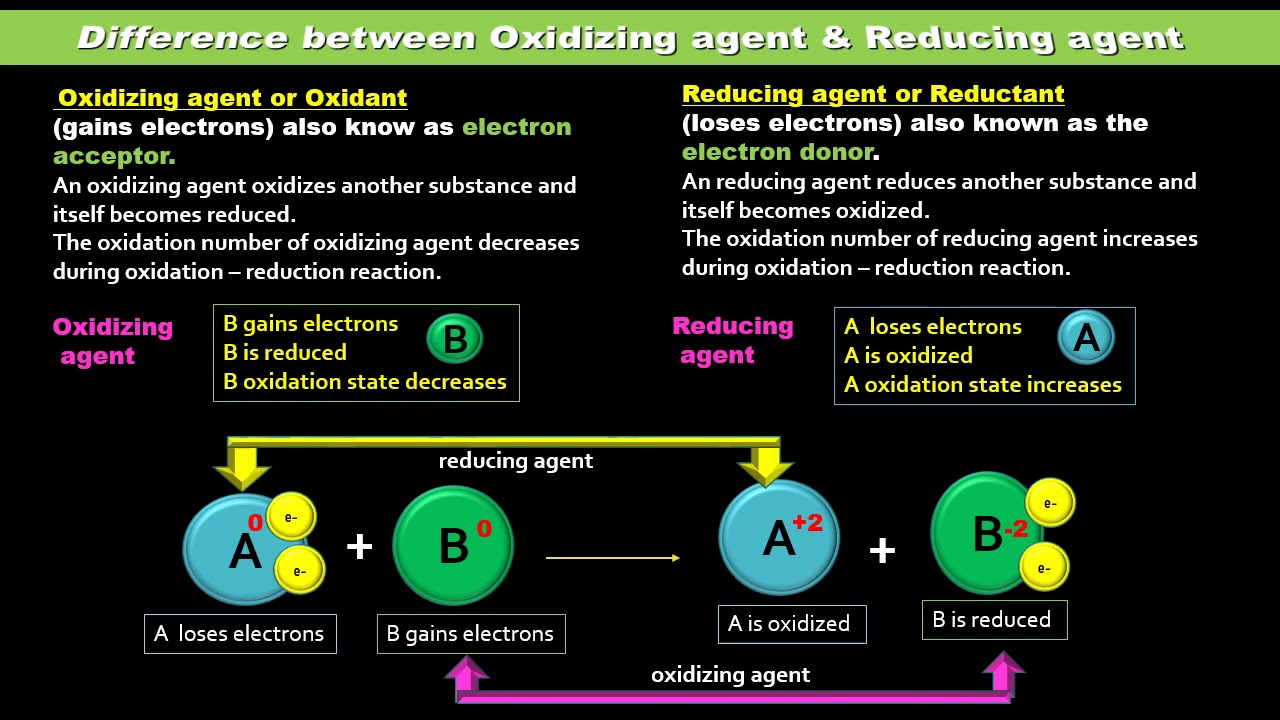
Oxidizing And Reducing Agents Reducing Agent Redox Reactions Chemistry

Oxidising Agent Or Oxidant Oxidizing Agent Chemistry Oxidation
No comments for "Which of the Following Is an Oxidizing Agent"
Post a Comment